Pentosan Injections for Osteoarthritis: Evidence, Safety and Why We No Longer Prescribe Them
Clinical Summary from Dr David Samra
Dr David Samra is a Fellowship-trained Sport and Exercise Medicine Physician with clinical experience treating osteoarthritis and bone marrow lesions in both athletes and the general population.
Pentosan polysulfate injections have generated interest as a potential treatment for osteoarthritis, particularly in patients with bone marrow lesions. While early studies have suggested potential benefits, the medication is currently not approved by the Therapeutic Goods Administration (TGA) for osteoarthritis in Australia.
After reviewing the available evidence and considering the regulatory and medico-legal context, Dr Samra does not currently prescribe pentosan injections. Patients who enquire about pentosan receive an evidence-based explanation of the current research and alternative treatment strategies.
Last reviewed: March 2026
Author Dr David Samra, MBBS (Hons), MD, FACSEP – Sport and Exercise Medicine Physician
Pentosan polysulfate (PPS) injections have attracted considerable interest as a potential treatment for knee osteoarthritis and bone marrow lesions.
Many patients search online for information about pentosan injections because early research has suggested the drug may improve joint pain and possibly influence the underlying disease process.
At Progressive Sports Medicine, we frequently receive enquiries about pentosan injections.
However, Dr David Samra does not currently prescribe pentosan injections.
This page explains:
- what pentosan is
- how it may work
- the current clinical evidence
- the regulatory status in Australia
- the potential safety considerations
- why we have chosen not to prescribe it at this time
Our goal is to provide balanced, evidence-based information so patients can make informed decisions.
What is Pentosan?
Pentosan polysulfate sodium (PPS) is a semi-synthetic polysaccharide derived from plant xylans.
It has been used in medicine for more than 60 years.
The oral form of PPS (brand name Elmiron) is approved in several countries for the treatment of interstitial cystitis, a chronic inflammatory bladder condition.
Researchers later discovered that PPS may have several biological effects relevant to osteoarthritis, including:
- anti-inflammatory activity
- inhibition of cartilage-degrading enzymes
- stimulation of cartilage matrix synthesis
- improvement in blood flow within the subchondral bone
- reduction of bone marrow lesions
Because of these mechanisms, PPS has been investigated as a potential disease-modifying osteoarthritis drug (DMOAD).
Reference
Ghosh P. The role of pentosan polysulfate in osteoarthritis.
Why Pentosan Generated so much Interest in Osteoarthritis
Osteoarthritis is one of the most common causes of pain and disability worldwide.
Most available treatments focus on reducing pain, but they do not significantly alter the underlying disease process.
Examples include:
- anti-inflammatory medications (NSAIDs)
- corticosteroid injections
- hyaluronic acid injections
- analgesic medications
These therapies can improve symptoms but do not slow cartilage degeneration.
For decades researchers have searched for disease-modifying osteoarthritis drugs (DMOADs).
A true DMOAD would ideally:
- reduce joint pain
- improve function
- slow cartilage loss
- reduce structural damage in the joint
To date, no drug has EVER conclusively achieved regulatory approval as a disease-modifying osteoarthritis therapy.
Reference
Karsdal MA et al. Disease-modifying treatments for osteoarthritis.
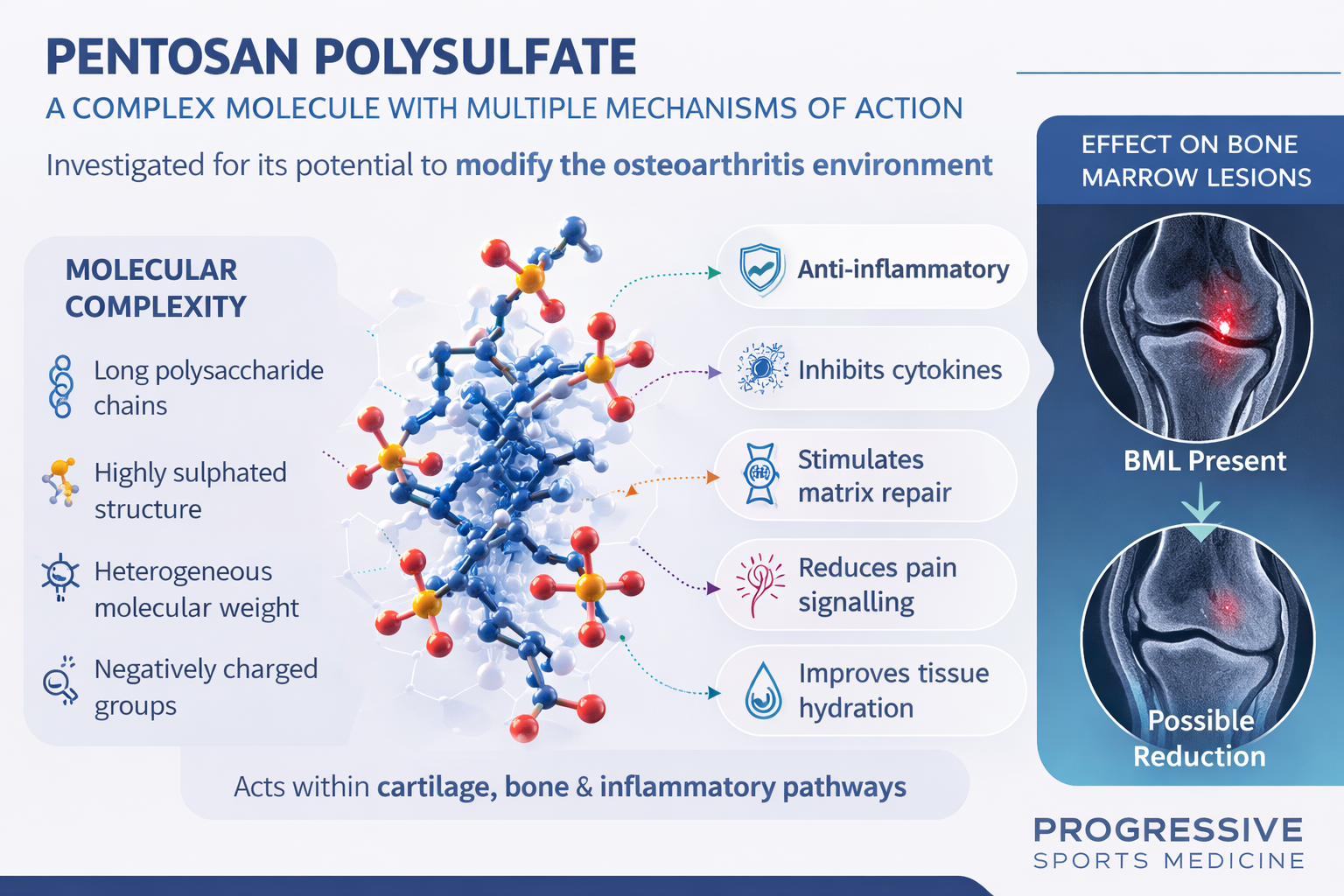
Bone Marrow Lesions in OA
Much of the interest in pentosan relates to its potential effects on bone marrow lesions (BMLs).
Bone marrow lesions are areas of inflammation and micro-damage in the bone beneath the cartilage.
They are commonly seen on MRI scans in patients with osteoarthritis.
Research has shown that:
- bone marrow lesions correlate strongly with pain
- large lesions increase the risk of cartilage loss
- progression of BMLs predicts worsening osteoarthritis
Because pentosan appears to improve blood flow and bone metabolism, it has been proposed that the drug may reduce these lesions.
Reference
Hunter DJ et al. Bone marrow lesions and osteoarthritis progression.
How Pentosan (iPPS) is Administered
When used experimentally for osteoarthritis, PPS is usually given as a subcutaneous injection.
The typical protocol used in early research involved:
two injections per week for six weeks
These injections are administered under the skin, similar to insulin injections.
Importantly:
the medication is not injected into the joint.
This means pentosan acts as a systemic therapy, affecting the entire body rather than a single joint.
This observation has contributed to growing interest in systemic factors in osteoarthritis, including inflammation, metabolic health, and vascular changes within bone.
Evidence for Pentosan in Osteoarthritis
Several early studies have investigated pentosan for osteoarthritis.
These studies suggest the drug may improve:
- knee pain
- joint function
- inflammatory biomarkers
- bone marrow lesions on MRI
Some patients reported meaningful improvements in activity levels and pain reduction.
However, it is important to understand that most of these studies were relatively small or early-phase trials.
Large Phase 3 trials, which are required for regulatory approval, are still ongoing.
Reference
Smith MM et al. Pentosan polysulfate for osteoarthritis: experimental and clinical evidence.
Current Regulatory Status in Australia
Pentosan injections for osteoarthritis are not approved by the Therapeutic Goods Administration (TGA).
Because of this, the medication can only be prescribed through the Special Access Scheme (SAS).
The SAS pathway allows doctors to prescribe medications that:
- are not yet approved in Australia
- have emerging but incomplete evidence
- may help patients when conventional treatments have failed
However, SAS prescribing does not replace the need for formal clinical trials required for full regulatory approval.
Safety Considerations
Like all medications, pentosan has potential side effects.
Known safety considerations include:
- mild anticoagulant effects (increased bruising or bleeding risk)
- gastrointestinal symptoms
- headache
- fatigue
More recently, long-term oral PPS exposure has been associated with a rare condition known as pigmentary maculopathy, affecting the retina.
Although this complication appears related to long-term high-dose oral exposure, the finding has increased regulatory caution.
Reference
Hanif AM et al. Pentosan polysulfate maculopathy.
Because pentosan is currently an off-label therapy, doctors who prescribe it must monitor patients closely for any potential complications.
Why Dr Samra No Longer Prescribes Pentosan
Dr David Samra previously prescribed pentosan through the TGA Special Access Scheme in carefully selected patients.
However, after reviewing the evolving evidence and regulatory landscape, he has chosen not to prescribe pentosan injections at this time.
This decision reflects several considerations:
1. Regulatory status
Pentosan injections remain unapproved for osteoarthritis in Australia.
While SAS prescribing is permitted, it carries additional medico-legal responsibilities.
2. Ongoing clinical trials
Large clinical trials are still underway to determine whether pentosan provides meaningful benefits compared with placebo.
As with all other OA treatments, it is entirely possible that the medication improves symptoms and function, without having any long-term, disease modifying effect.
Until these results are available, the long-term benefits remain uncertain.
3. Safety monitoring
Because pentosan is an off-label systemic therapy, careful monitoring is required.
For patients travelling long distances or seeking treatment from interstate, adequate follow-up can be difficult.
4. Evidence-based practice
At Progressive Sports Medicine we prioritise treatments that are supported by strong clinical evidence and established regulatory approval.
Evidence-Based Osteoarthritis Treatment
While there is ongoing research into new medications, the most effective osteoarthritis treatments remain:
Exercise therapy
Strengthening muscles around the joint improves pain and function.
Weight management
Reducing body weight decreases mechanical load on the joint.
Metabolic health
Systemic inflammation and metabolic factors influence osteoarthritis progression.
Activity modification
Adjusting training loads can help protect joints while maintaining activity.
Injection therapies
In some patients, treatments such as:
- corticosteroid injections
- platelet-rich plasma (PRP)
- hyaluronic acid
may provide temporary symptom relief.
Importantly, these treatments should be considered as part of a comprehensive management strategy, not in isolation.
The Future of Pentosan Research
Research into pentosan continues.
Several ongoing trials (including large-scale, well-designed phase 3 trials) aim to determine whether PPS can:
- improve pain and function
- reduce bone marrow lesions
- slow structural progression of osteoarthritis (this is the hardest to prove but it is what all parties are hoping will be established)
If future studies confirm these benefits with an acceptable safety profile, pentosan may eventually become an approved treatment.
However, until high-quality evidence is available and regulatory approval is granted, it remains an investigational therapy.
Key Takeaways
- Pentosan polysulfate (PPS) is an older drug being investigated for osteoarthritis.
- Early studies suggest potential benefits for pain and bone marrow lesions.
- Large clinical trials are still underway.
- Pentosan injections are not currently approved by the TGA for osteoarthritis.
- Dr David Samra does not currently prescribe pentosan injections.
Patients seeking treatment for osteoarthritis should focus on evidence-based management strategies that improve joint health, strength, and long-term function.
Specialist Osteoarthritis Assessment
If you are experiencing persistent joint pain or osteoarthritis, a detailed assessment can help determine the most appropriate treatment options.
At Progressive Sports Medicine, we provide comprehensive evaluation of:
- joint structure
- biomechanics
- muscle strength
- metabolic health
to develop an individualised management plan.
Medical Disclaimer
This article is intended for educational purposes only and does not constitute medical advice. Individual treatment decisions should always be discussed with a qualified healthcare professional. Progressive Sports Medicine has no commercial relationships with pharmaceutical companies or manufacturers of pentosan products.
Scientific References:
Ghosh, P., Smith, M. and Wells, C. (2005) Pentosan polysulfate in osteoarthritis. Arthritis Research & Therapy, 7(3), pp. R495–R500.
Karsdal, M.A., Michaelis, M., Ladel, C., Siebuhr, A.S., Bihlet, A.R., Andersen, J.R., Guehring, H., Christiansen, C., Bay-Jensen, A.C. and Kraus, V.B. (2016) Disease-modifying treatments for osteoarthritis (DMOADs) of the knee and hip: lessons learned from failures and opportunities for the future. Osteoarthritis and Cartilage, 24(12), pp. 2013–2021.
Hunter, D.J., Zhang, Y., Niu, J., Goggins, J., Amin, S., Guermazi, A., Gale, D., Felson, D.T. (2011) The association of bone marrow lesions with pain and structural progression in knee osteoarthritis. Nature Reviews Rheumatology, 7(4), pp. 213–222.



